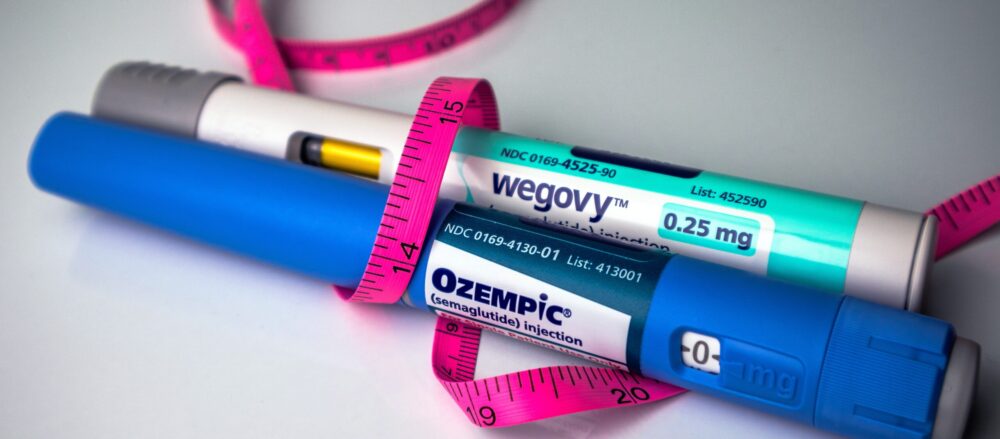
A move by the U.S. Food and Drug Administration could significantly change how millions of Americans access some of the most widely used weight loss medications on the market. On April 30, the FDA announced a proposal to remove glucagon-like peptide-1 (GLP-1) receptor agonists including semaglutide and tirzepatide, the active ingredients in Ozempic, Wegovy and Mounjaro from what is known as the 503B bulks list.
That list governs which drug substances outsourcing facilities are permitted to compound in large quantities without individual prescriptions. If the proposal moves forward, it would cut off the primary channel through which many patients currently obtain lower-cost versions of these in-demand medications.
What the 503B bulks list actually is
To understand the stakes, it helps to know what the 503B bulks list does. Under section 503B of the Federal Food, Drug, and Cosmetic Act, specialized outsourcing facilities are allowed to produce compounded medications in bulk meaning they can prepare large batches of drugs without waiting for individual patient prescriptions. These facilities operate under FDA oversight, but the compounded products they produce do not go through the same pre market approval process required of brand name drugs.
The key difference, according to bariatric medicine specialists, is the level of scrutiny involved. FDA approved medications must clear extensive clinical trials before reaching patients, while compounded versions do not face the same threshold of review, which is precisely why regulators are now questioning their continued availability at scale.
Why this proposal worries doctors and patients
Most compounded GLP-1 medications currently reaching patients come from 503B outsourcing facilities. If those facilities are no longer permitted to compound these drugs in bulk, availability would drop sharply. While 503A compounding pharmacies the traditional, smaller scale option could still prepare the drugs for individual patients, that pathway is far more limited and not built to meet widespread demand.
Medical professionals working in bariatric care have raised concerns that patients cut off from compounded options may turn to unregulated sources outside the United States to fill the gap. That scenario carries its own serious risks, including contaminated formulations, incorrect dosing and a complete absence of quality control. Several medical societies have already recommended against compounded GLP-1 therapies, citing inconsistent quality and a higher rate of severe side effects compared to their FDA approved counterparts.
Safety questions that were already there
Even before this proposal, the safety of compounded GLP-1s was a subject of debate in the medical community. The quality of these medications can vary widely from one facility to another. While some compounding pharmacies maintain rigorous internal standards, the absence of FDA approval leaves room for contamination, mislabeling and dosing errors that can cause serious harm.
Research has suggested that compounded versions of these drugs are associated with a greater number of severe adverse events than the commercially approved products a data point that appears to be driving the FDA’s current thinking.
What patients can do right now
For those currently on compounded GLP-1 therapy, medical experts recommend having a direct conversation with a healthcare provider before any changes are forced. The proposal is not yet final, and patients should not make any medication decisions without professional guidance.
In the meantime, several paths forward exist. There are FDA approved weight management medications on the market, and structured medical weight loss programs that combine nutritional guidance, behavioral support and clinical monitoring remain a proven option.
For patients concerned about cost, pharmaceutical companies that manufacture brand name GLP-1 drugs often offer copay assistance cards and patient assistance programs that can dramatically reduce out of pocket expenses. Prescription savings platforms can also provide meaningful discounts for those without insurance coverage or whose plans do not yet include these medications.
Insurance formularies are worth revisiting as well coverage policies for weight loss drugs have been shifting, and what was excluded a year ago may now be covered.
The bigger picture
The FDA’s proposal does not eliminate GLP-1 medications it targets the lower cost, compounded versions that have made these treatments accessible to a broader group of patients. As the regulatory process continues, those who depend on these drugs will need clear communication from their providers and a backup plan ready to go.